Treating hazardous waste in the country of origin prior to exporting overseas can be extremely risky because of the numerous conflicting state, federal, and international laws.
Don’t Landfill. Reclaim
shipping of spent catalyst for reuse.
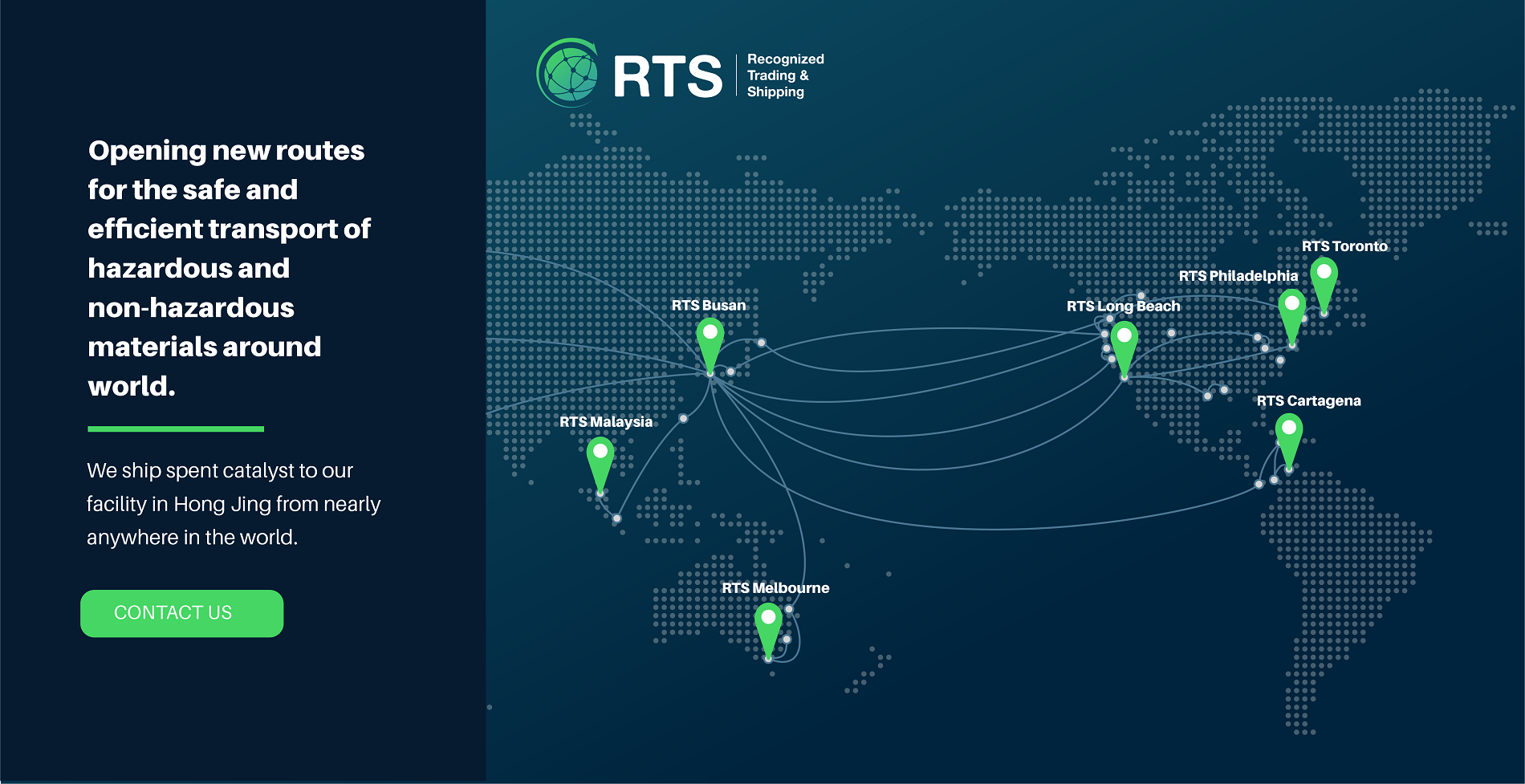
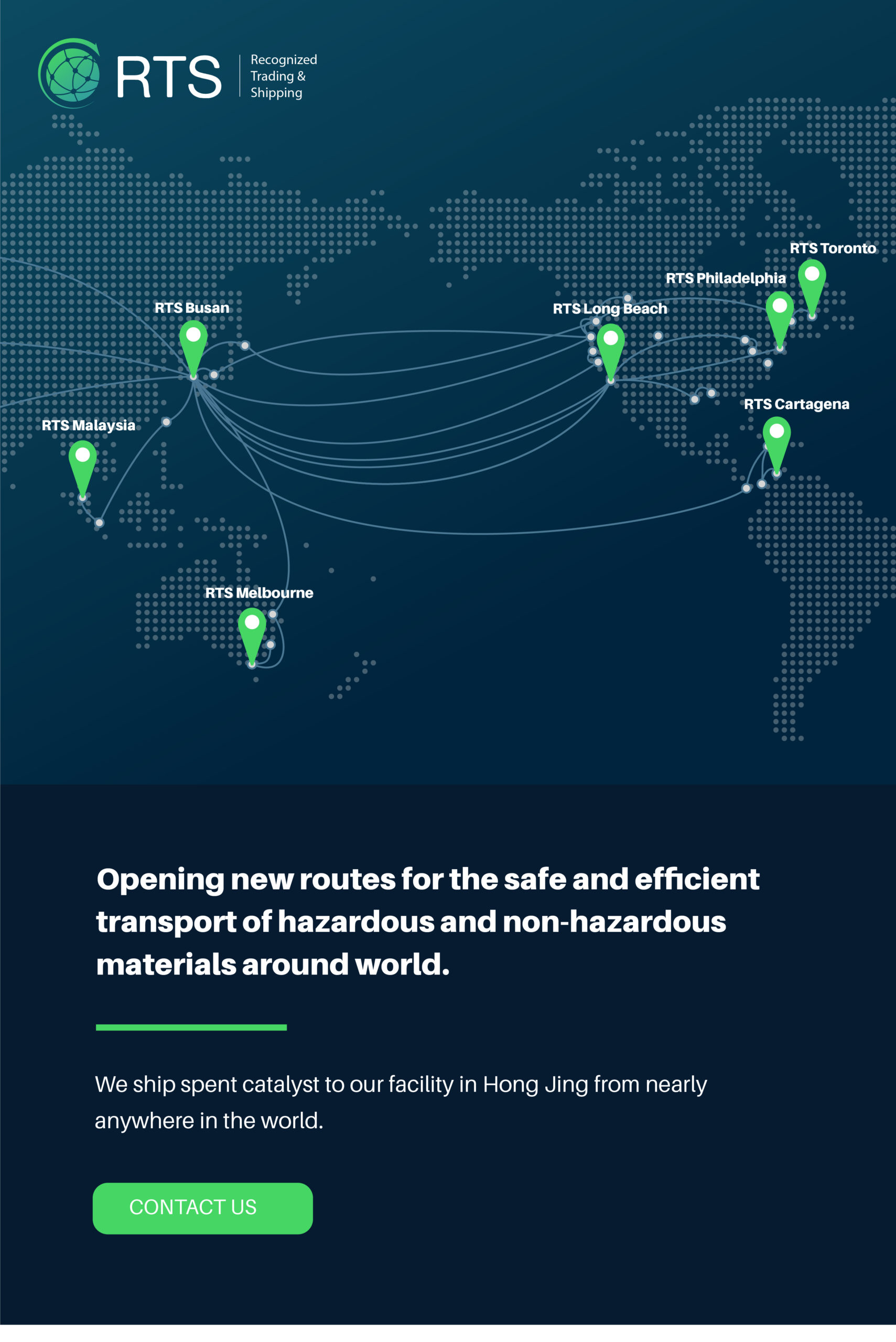
We’re changing the way hazardous and non-hazardous waste are transported
RTS provides a complete solution for the movement of hazardous and non-hazardous waste across international boundaries. Through long standing industry connections and sector leading knowledge of EPA Guidelines, we set the standard for shipping spent catalyst around the world.
A Strong Network of Global Partnerships

Global shipping today is more complicated than ever. Because of our dedicated knowledge of international regulations and commitment to complete transparency, RTS has built a strong network of partners that allows us to ship almost anywhere in the world.
- A to Z handling and shipping of hazardous waste
- 20+ years of experience in the industry
- Guaranteed compliance with OECD, Basel Convention, 40CFR, Title 22, and the Waste German Act
- EPA partner
- Trusted by the world’s largest exporters of spent catalyst of Hazardous Waste and Renewable Energy Resources
Our Partners






No more bin rental agreements or late fees.

Our waste packaging options make it easy for our customers to ship with no added headaches or hassles.
- No more late fees or past due bills
- Hassle-free and safe transportation
- No more complex procedures and roadblocks
The Safest and Fastest Shipping of Hazardous Waste at the Best Price.
GET A QUOTE:Articles and Resources
Transporting secondary raw materials between countries is becoming increasingly common and requires a deep knowledge of material classifications and international environmental agreements.
Reaching global climate neutrality by 2050 is an ambitious goal and one that requires the cooperation of oil companies.
Treating hazardous waste in the country of origin prior to exporting overseas can be extremely risky because of the numerous conflicting state, federal, and international laws.
Transporting secondary raw materials between countries is becoming increasingly common and requires a deep knowledge of material classifications and international environmental agreements.
Reaching global climate neutrality by 2050 is an ambitious goal and one that requires the cooperation of oil companies.
Working with experienced waste disposal companies will help protect the environment, strengthen your brand’s reputation, and ensure the safety of your employees, and that of people around the world.
Locations
USA - Global Headquarters
217 Rosecrans Avenue
Manhattan Beach, CA 90266 - United States
Busan, Korea
7F PanOcean Bldg., 102 Jungang Daero
Jung-gu, Busan - South Korea
Toronto, Canada
330 Bay St. Unit 1400
Toronto, Ontario M5H 2S8 - Canada
London, England
483 Green Lanes
London, LDN N13 4BS - United Kingdom
Melbourne, Australia
Level 8, 230 Collins Street
Melbourne, Vic 3000 - Australia
USA: 1 (213) 444-9966
KOR: 82 (51) 998-6572
CAN: 1 (416) 512-3771
UK: 44 (020) 8263-0867
AUS: 61 (03) 7019-4222
Email: info@recognizedtrader.us



